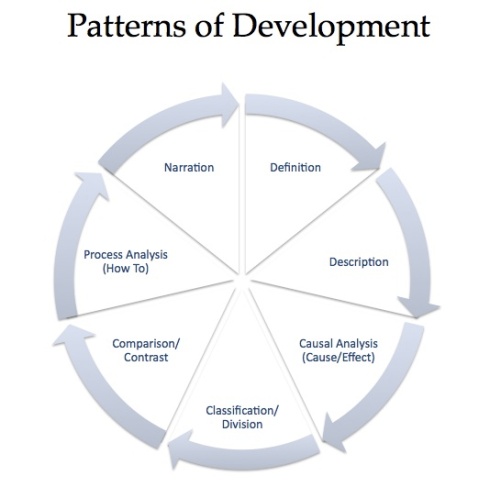
Ionic equations and spectator ions - BBC Bitesize.
Hi. I need help with solving this question. Write the net ionic equation for the neutralization reaction of H3PO4 (aq) with Ba(OH)2(aq) Thanks.
The net ionic equation is a chemical equation for a reaction that lists only those species participating in the reaction. The net ionic equation is commonly used in acid-base neutralization reactions, double displacement reactions, and redox reactions. In other words, the net ionic equation applies to reactions that are strong electrolytes in.
How to create ionic equations for neutralisation, precipitation and redox reactions from word or chemical equations. Read more. Free. Loading. Save for later. Preview and details Files included (1) pptx, 641 KB. Ionic Equations. About this resource. Info. Created: Nov 17, 2014. pptx, 641 KB. Ionic Equations. Report a problem. This resource is designed for UK teachers. View US version.
The ionic equation represents the 'actual' chemical change and omits the spectator ions. Five types of examples of ionic equations are presented below including neutralisation, salt precipitation and redox equations. (1) Acid-base neutralisation reactions: Acids can be defined as proton donors.
Many can involve ionic species in these reactions, but in some cases we find that there are ionic species that are unchanged appearing on both the reactant and product sides of a reaction equation.
Hi I was just doing my Chemistry revision for my A-lvl exam on January and got kinda stuck writing ionic equation. So i was thinking if its possible t.
Neutralization reactions occur when an acid reacts with a base to produce a salt and water. This is a double replacement reaction because the ions in compounds HX and MOH switch partners. Therefore, neutralization reactions are double replacement reactions. The driving force behind neutralization reactions is the formation of water. An acid reacts with a base to produce a salt and water.